 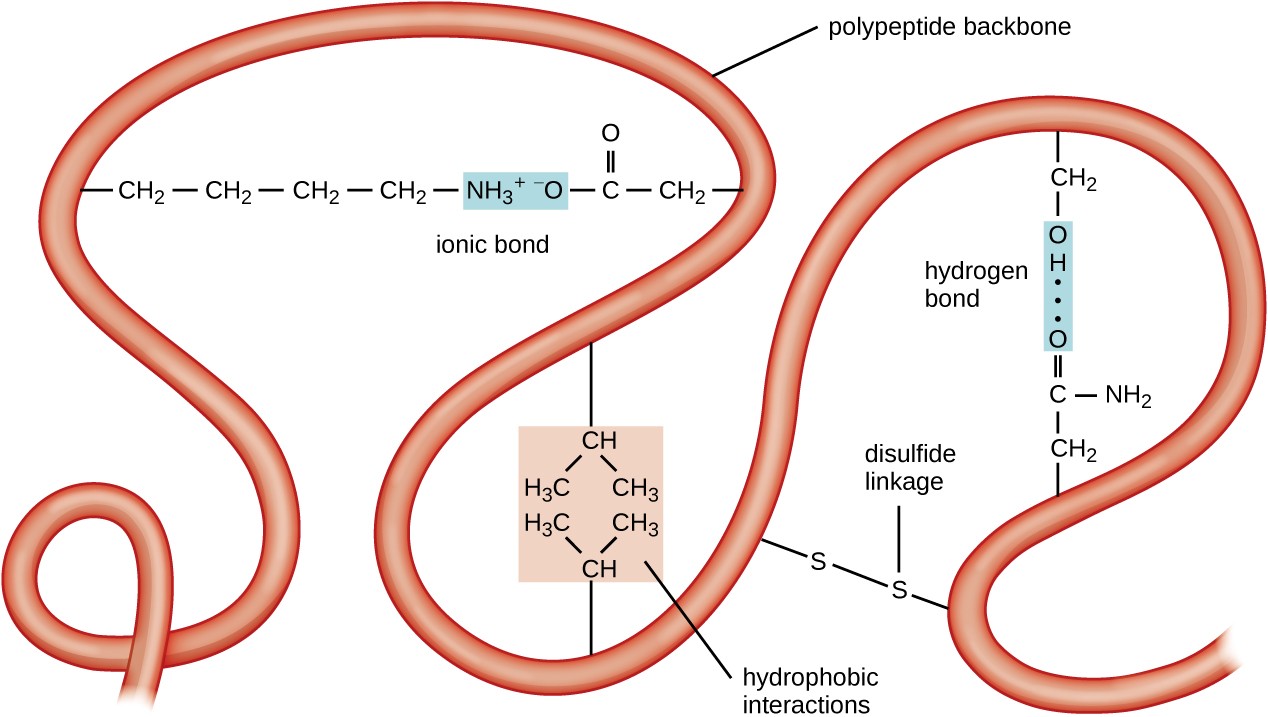
Because of the partial double bond between the α carbon and the amine nitrogen, no rotation is possible around that bond. The four atoms that are part of the peptide bond are shown as larger spacefilling models. The amino acids are taken from the crystal structure of hemoglobin The structure at the right shows a peptide bond between the amino acids valine (Val) and serine (Ser). Thus, there is no rotation around the bond.Ĭlick on the structure below to switch the resonance forms of the peptide bond. This means that the peptide bond (the C=O and N-H) all reside in a single plane. Also note the first peptide chain possesses an internal loop.Interestingly, peptide bonds have a second resonance form, as demonstrated below. He found the primary structure to comprise of two chains linked by two cysteine disulfide bridges. However, these hydrogen bonds between the atoms of the polypeptide backbone do not discriminate between the various residues involved since the backbone. In PDB format, a standard nomenclature is used for standard amino acid and nucleic acid residues. This pioneering work, completed in 1953 after some 10 years of effort, earned a Nobel Prize for British biochemist Frederick Sanger (born 1918). Chain IDs, residue names and numbers, and atom names are read from the input file. Insulin was the first protein whose amino acid sequence was determined. Remember that reduction is the addition of hydrogen.Ĭysteine residues in the the peptide chain can form a loop buy forming the disulfide bond (-S-S-), while cysteine residues in different peptide chains can actually link what were otherwise separate chains. The reduction of a disulfide bond is the opposite reaction which again leads to two separate cysteine molecules. This is an important bond to recognize in protein tertiary structure. Of the many backbone modifications that render a peptide bond recalcitrant to proteolysis 10, 11 we found methylation of the amide nitrogen particularly useful, since N-methylated peptides can easily be obtained by solid phase custom synthesis. The sulfurs (yellow) join to make the disulfide bridge. its flexibility, determines the properties of the polymer (such as the glass transition temperature). Polymers are often classified according to the elements in the main chains. The peptide backbone consists of repeating units of 'N-H 2, CH, C double bond O N-H 2, CH, C double bond O etc. 1 In polymer science, the polymer chain or simply backbone of a polymer is the main chain of a polymer. An unspecified oxidizing agent (O) provides an oxygen which reacts with the hydrogen (red) on the -SH group to form water. The structure of a peptide can be written fairly easily without showing the complete amide synthesis reaction by learning the structure of the 'backbone' for peptides and proteins. The oxidation of two cysteine amino acids is shown in the graphic. 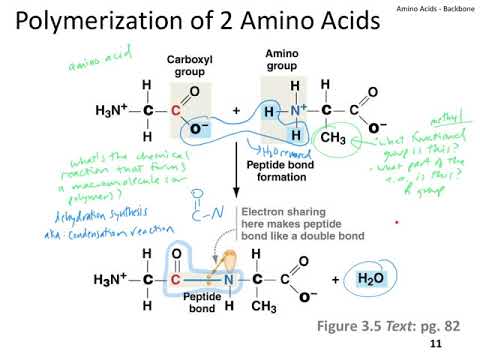
The oxidation of two sulfhydryl groups results in the formation of a disulfide bond by the removal of two hydrogens. The amino acid cysteine undergoes oxidation and reduction reactions involving the -SH (sulfhydryl group). In organic chemistry, a peptide bond is an amide type of covalent chemical bond linking two consecutive alpha-amino acids from C1 (carbon number one) of one. the extent of interactions with the backbone nitrogen atom, as long as. An important resonance contributor has a C=N double bond and a C-O single bond, with a separation of charge between the oxygen and the nitrogen.Īlthough B is a minor contributor due to the separation of charges, it is still very relevant in terms of peptide and protein structure – our proteins would simply not fold up properly if there was free rotation about the peptide C-N bond.ĭisulfide Bridges and Oxidation-Reduction and the peptide backbone nitrogen (N), which is the only hydrogen bond donor. This, along with the observation that the bonding around the peptide nitrogen has trigonal planar geometry, strongly suggests that the nitrogen is sp 2-hybridized. One of the most important examples of amide groups in nature is the ‘peptide bond’ that links amino acids to form polypeptides and proteins.Ĭritical to the structure of proteins is the fact that, although it is conventionally drawn as a single bond, the C-N bond in a peptide linkage has a significant barrier to rotation, almost as if it were a double bond. Resonance contributors for the peptide bondsĪ consideration of resonance contributors is crucial to any discussion of the amide functional group. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
